Дуркабтаген аутолейцел
Durcabtagene autoleucelМНН
Rec. INN (наименование, зарегистрированное ВОЗ)
Химическое название
autologous T lymphocytes obtained from peripheral blood mononuclear cells by leukapheresis, transduced with a self-inactivating, non-replicating lentiviral vector encoding a chimeric antigen receptor (CAR) targeting human B cell maturation antigen (BCMA). The expressed transgene comprises a CD8α leader sequence, an anti-BCMA single chain variable fragment (scFv), a CD8α hinge and transmembrane domain, and a 4-1BB (CD137) and CD3ζ signalling domain, under control of the human elongation factor 1α (EF-1α) promoter. The construct is flanked by 5' and 3' long terminal repeats (LTRs) and also contains a ψ packaging signal, a truncated gag, a Rev response element (RRE), a central polypurine tract (cPPT) sequence and a synthetic ID tag sequence. The leukapheresis material is enriched for CD4+ and CD8+ T lymphocytes by positive immunoselection, activated by CD3 and CD28 agonists and transduced with the lentiviral vector in serum-free media containing interleukin 2 (IL-2). Without further cell propagation in vitro, the T lymphocytes are washed, formulated for infusion, and cryopreserved. The T lymphocytes phenotype very closely resembles that of the initial leukapheresis material. The cell suspension consists of T lymphocytes (≥80%; ≤1% B cells), with a minimum of 4.0% of the T lymphocytes expressing the CAR-BCMA transgene. The T lymphocytes secrete interferon gamma (IFN-γ) and IL-2 following co-culture with BCMA- expressing cells
Структура
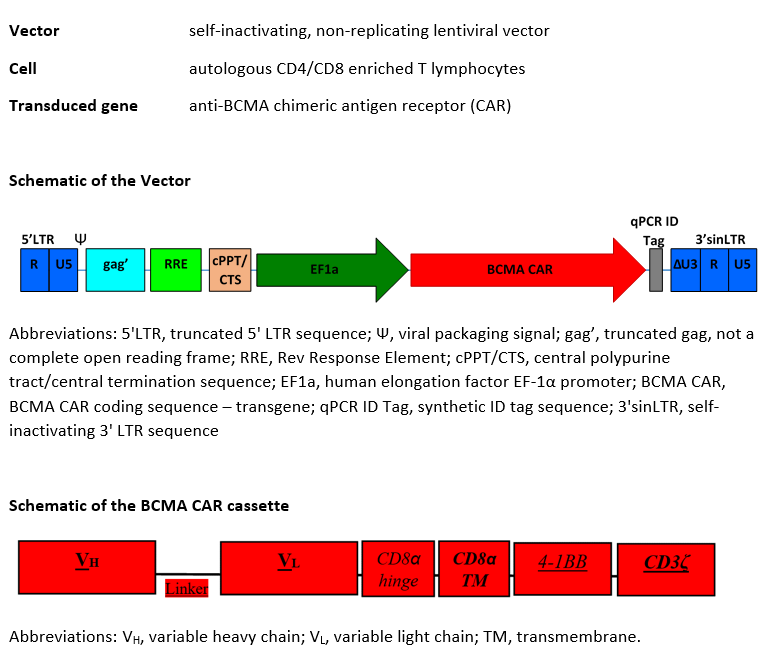
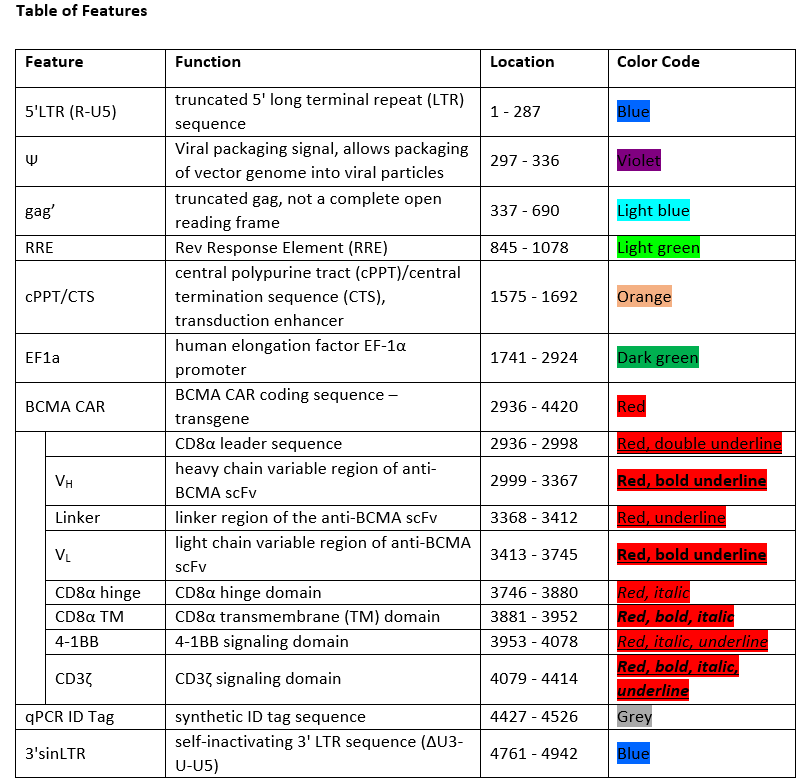


Иностранные названия
- Durcabtagenum autoleucelum (латинское)
- Durcabtagene autoleucel (английское)
- Durcabtagen autoleucel (немецкое)
- Durcabtagène autoleucel (французское)
- Durcabtagén autoleucel (испанское)
Подробнее по теме
Узнайте дополнительные сведения о действующем веществе Дуркабтаген аутолейцел:
